
Liposomal dietary supplements: what is the difference between liquid and powder forms?

Liposomal dietary supplements have become one of the most discussed technologies in the dietary supplement world in recent years.
Liposomal technology has become one of the most widely used terms in the world of dietary supplements in recent years. More and more products described as liposomal are appearing on the market, from vitamin C to B vitamins to plant extracts and low-solubility compounds.
At the same time, we are increasingly seeing two different technological approaches:
- liquid liposomal formulations,
- powders referred to as liposomal.
At first glance, the two solutions may appear similar. In practice, however, the difference between the two is significant – both from the point of view of technology and the subsequent quality of the product.
So it’s worth asking a basic question: does every supplement described as liposomal work in the same way?
What is a liposome? Short structure
A liposome is a microscopic structure made up of phospholipids, or molecules that have two distinct parts:
- The hydrophilic, or “water-loving,” part,
- The lipophilic, or “fat-loving,” part.
Thus, phospholipids in an aqueous environment spontaneously arrange themselves into a double lipid layer. A small vesicle is then formed, with an aqueous phase in the center and a phospholipid membrane as the outer boundary.
It is this structure that is called a liposome.
In practice, this means that a liposome can act as a carrier for various substances:
- hydrophilic that can be found in the aqueous interior,
- Lipophilic compounds that can bind to the lipid layer.
This solution is not something detached from nature. Lipid systems are well known to the body because they resemble cell membranes in structure. This is why liposomal technology is attracting so much interest in the area of supplements and formulations of bioactive ingredients.
Why liposomes increase bioavailability
The popularity of liposomes is due to one key reason: they can improve the bioavailability of active ingredients.
Bioavailability is, in simple terms, the degree to which an ingredient is absorbed and becomes available to the body. The mere presence of a substance in a capsule, powder or liquid does not yet mean that the body will actually use it.
In the case of a well-designed liposomal formulation, benefits can arise from several mechanisms:
- Protecting the active ingredient from partial degradation,
- easier contact with biological membranes,
- better dispersion in an aqueous environment,
- Increase contact area due to small particle size.
This is especially important with ingredients that have poor solubility or limited absorption in classic form.
Two main liposomal technologies on the market
Although many products use the same marketing term, in practice there are two different technological models in the market.
Liquid liposomal formulations
In this case, the liposomes or lipid dispersion already exist in the finished product. This means that the manufacturer prepares the system in the aqueous phase and stabilizes it before it is administered to the customer.
It is such a product that can realistically be tested as a finished formulation.
Powders described as liposomal
In this group, we usually do not deal with ready-made liposomes present in the aqueous phase. It is usually a dry system containing:
- active ingredient,
- phospholipids,
- sometimes excipients.
When water is added or after contact with gastrointestinal fluids, such a powder can form lipid structures that are designed to resemble a liposomal system.
And this is where the most important difference comes in:
in a liquid product, the quality of the layout can be assessed right away, while in a powder the actual layout is only created later.
How liquid liposomes are made
The liquid liposomal formulation is created in an aqueous environment. Depending on the technology, appropriately selected ones are used:
- phospholipids,
- aqueous phase,
- mixing parameters,
- temperature,
- Homogenization or sonication energy.
The goal is to obtain as homogeneous a dispersion as possible with controlled particle size and adequate stability.
The biggest advantage of this approach is that the finished product can be realistically evaluated in a laboratory. You can check:
- average particle size,
- size distribution,
- system stability,
- batch repeatability.
In other words: the manufacturer does not rely solely on the assumption that the system will “form later,” but can study what actually reaches the user.
What are liposomal powders
Powder products described as liposomal are usually an intermediate form. In technological parlance, they can be referred to as dry phospholipid systems or proliposomes.
This does not automatically mean that they are worthless. Instead, it means that you need a very precise understanding of what they are.
Such a product usually includes:
- active ingredient,
- A phospholipid or mixture of phospholipids,
- Sometimes stabilizers or excipients.
After contact with water, this system can begin to organize itself into various lipid structures. The problem is that without a test, one does not know exactly:
- Whether liposomes are actually formed,
- What size they are,
- What part of the system is small particles and what part is larger aggregates,
- How stable such a dispersion is.
This is the most important point of the whole discussion.
What happens when you add water to liposomal powder
Many users assume that if a product is called liposomal, when water is added it will automatically produce a stable, well-organized liposome structure. Technologically, it is not that simple.
Once the powder is hydrated, different systems can be formed:
- small liposomes,
- larger liposomes,
- micelles,
- lipid aggregates,
- plain emulsion,
- A mixture of several populations of particles.
And that is why the mere declaration “liposomal powder” does not yet say much about the final quality of the system.
In practice, it all depends on:
- Quality of phospholipids,
- ingredient ratio,
- reconstitution method,
- water temperatures,
- mixing time,
- The stability of the system after creation.
If the manufacturer does not show post-hydration testing, the user is not sure what is really being produced.
.
Why particle size matters
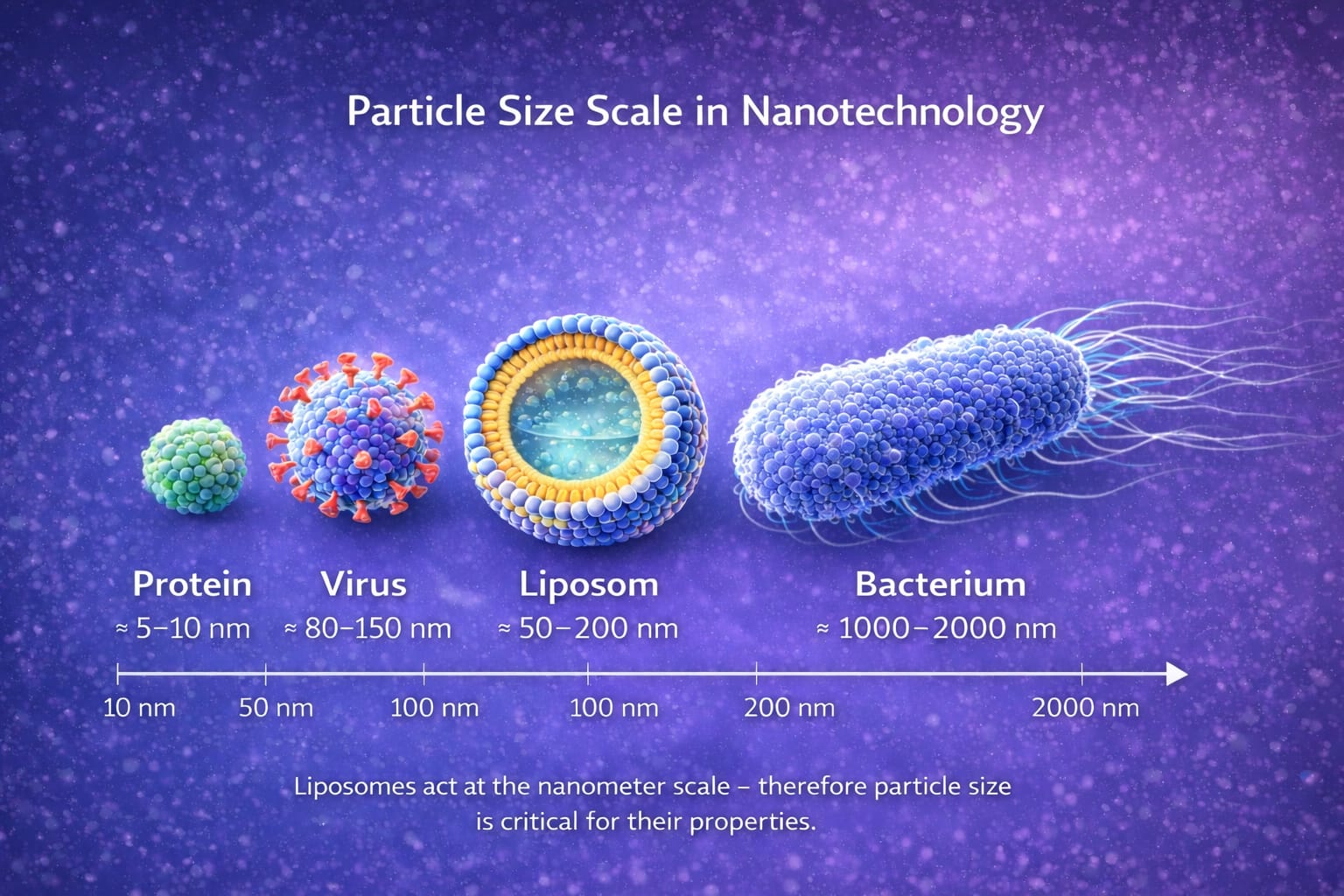
Fig.2 Comparison of the size of liposomes (50-200 nm) with other biological structures at the nanometer scale.
In liposomal and nanoemulsion technology, particle size is one of the most important features of the entire formulation. In the case of herbal extracts, you can also see large technological differences between classical forms and liposomal formulations.
We described this in more detail in the article “Liposomal herbal supplements – how do they differ from classical extracts?”.
It is the size of the lipid structures that largely determines the physical properties of the system and how it behaves in an aqueous environment.
In the case of liposomal formulations, we are usually talking about structures ranging in size from tens to hundreds of nanometers. This is a much smaller scale than most biological structures, such as cells or bacteria.
For a better understanding of this scale, it is worth noting that:
- A typical cell is several to tens of micrometers in size,
- Bacteria are usually around 1000-2000 nanometers,
- Viruses are often in the 80-150 nanometer range.
Liposomes are thus in the nanometer range, the scale of very small biological structures.
Particle surface and formulation properties
Reducing the size of the particles results in a very large increase in their total surface area.
If the same amount of substance is separated into a very large number of small structures instead of a few large aggregates, the total contact area with the environment becomes much larger.
In practice, this means, among other things:
- better dispersion in water,
- More stable dispersion of components,
- More homogeneous physical and chemical properties of the formulation.
This is why many pharmaceutical and nutraceutical technologies strive for the smallest yet stable particles possible.
The homogeneity of the system is equally important
Average particle size alone, however, does not tell all about the quality of the formulation.
Two samples can have a similar average particle size, for example, around 120 nm, and yet completely different internal structure.
In the first case, most particles can be very similar in size, forming a homogeneous lipid system.
In the second case, the same average value can result from the mix:
- very small lipid structures
- large fat aggregates
- irregular micellar structures
Such a system is much less stable and much more difficult to predict in terms of physical properties.
Particle size distribution
Therefore, in the analysis of liposomal technologies, not only the average particle size itself is extremely important, but also the distribution of particle size throughout the sample.
If most of the structures fall within a narrow size range, the system is more homogeneous and stable.
If, on the other hand, the distribution is broad and both very small particles and large lipid aggregates appear in the sample, the formulation may behave quite differently than the average number given in nanometers alone would suggest.
For this reason, technological analyses also very often evaluate a parameter that determines the homogeneity of the system, which allows to determine whether the lipid structures are similar in size.
The importance of particle size in practice
In technological practice, differences in particle size can affect many formulation properties, including:
- The stability of the system over time,
- method of dispersion in water,
- resistance to lipid aggregation,
- Homogeneity of the entire dispersion.
Therefore, in the case of liposomal technologies, the mere declaration “liposomal” does not yet say much about the quality of the formulation.
Two products described in a similar way may have completely different technological properties precisely because of differences in the size and structure of the lipid particles.
Why laboratory tests are needed
Since lipid structures are nanometer-sized, they cannot be easily assessed visually.
In most cases, only appropriate analytical methods can determine:
- particle size,
- distribution of their size,
- Homogeneity of the entire system.
That’s why liposomal technology research uses specialized laboratory methods that allow particles to be analyzed at the nanometer scale.
How liposomes are tested in practice
One of the most commonly used tests in evaluating such formulations is DLS, or Dynamic Light Scattering.

Fig.3 Schematic of particle size study in liposomal formulation using DLS (Dynamic Light Scattering) method
This study makes it possible to determine:
- average particle size,
- size distribution,
- The level of homogeneity of the system.
PDI (polydispersity index) is also often analyzed, an index that tells whether a sample contains similar-sized particles or a rather broad and chaotic mixture.
It is this kind of research that allows one to answer the question of whether a given system is indeed nano-lipid in nature, or is simply a mixture of larger structures.
And here we come to a very important conclusion:
inthe case of powder, the most important tests are performed after reconstitution, that is, after adding water.
Are all liposomal products the same
Definitely not.
Under the common word “liposomal” there can be very different technological solutions. The final result is influenced by, among other things:
- type of phospholipids,
- ingredient proportions,
- production method,
- degree of encapsulation,
- particle size,
- system stability.
Therefore, two products described by the same word can work quite differently.
One formulation may be a well-organized and stable dispersion with small particles, while another is merely a system that is theoretically supposed to form when water is added, but in practice yields wide distribution and large aggregates.
It’s not a question of the name itself. It’s a question of the quality of the technology.
Is liposomal powder the same as liquid liposome ?
Don’t.
This is the shortest and fairest answer.
The liquid liposomal system is a finished formulation in an aqueous environment that can be tested and characterized even before use.
Liposomal powder is most often a dry phospholipid system that can only form lipid structures after contact with water. The final result therefore depends on the reconstitution process and the quality of the technology itself.
This does not mean that every powder is bad by definition. It does mean, however, that it requires much more caution in evaluation.
What to look for when choosing a liposomal supplement
If we want to look at the product more consciously, it is worth noting a few things.
1. does the manufacturer describe the technology in specific terms
Generic marketing slogans are not enough. The more specific, the better.
2. are particle size tests available
This is one of the most important parameters for lipid formulations.
3. is it known what the system looks like after reconstitution
This is especially important with powders.
4. what phospholipids were used
The quality of the lipid source is of great importance.
5. whether the product is stable
Stability is not a marketing add-on, but one of the basic qualities of quality.
What this means in practice for the user
The most important conclusion is simple:
not every form described as liposomal necessarily implies the same level of technological advancement.
In the world of supplements, it is worth distinguishing:
- finished lipid system in the product,
from - a system that is yet to emerge after use.
This distinction is crucial if we want to evaluate the real quality of a product, not just its name.
Liposomal plant supplements – a new direction of development
The most interesting direction in the development of liposomal technology today concerns not only classic vitamins, but also plant compounds with low solubility and limited bioavailability.
It is in this area that formulation often matters most. This is especially true for substances that are difficult for the body to use effectively in their usual form.
In such cases, technology is not just a marketing add-on. It becomes a viable formulation tool.
Summary
Liposomal technology is one of the most interesting developments in modern supplements. However, it is worth looking at it with precision.
The most important difference between liquid and powder form is this:
- In the liquid formulation, the lipid system already exists in the finished product,
- in powder, the system is yet to be formed after contact with water.
And this means that in the case of powder, tests performed after reconstitution – especially particle size and stability analysis – are crucial.
The more developed the technology, the less room is left for guesswork and more for realistic formulation parameters.
The most common questions about liposomal supplements (FAQ)
Does every supplement labeled as liposomal work the same way?
Don’t. The term “liposomal” refers to the method of formulation, but in practice the technological differences between products can be very large. The end result is affected by the quality of the phospholipids, particle size, stability of the system and the method of production, among other factors. Therefore, two products described as liposomal may differ significantly in physical properties and behavior in an aqueous environment.
Does liposomal powder always form liposomes when water is added?
Not always. In the case of many powder products, we are dealing with a dry mixture of ingredients, which can only form lipid structures after contact with water. However, the final effect depends on the quality of the technology, the proportions of the ingredients and the method of reconstitution. Therefore, in practice, it is important to evaluate such formulations also after adding water.
How can I check the quality of the liposomal formulation?
One of the most commonly used methods is DLS (Dynamic Light Scattering ) analysis. This test allows you to determine the size of the particles in the sample and their distribution. This allows you to see whether nano structures or larger aggregates dominate the preparation.
Why is particle size in liposomal formulations important?
Particle size has a major impact on the physical properties of the overall system. Smaller and more homogeneous particles usually mean a larger contact area and better dispersion stability. Therefore, in liposomal technology, particle size control is one of the basic quality parameters.
Are liposomes also found in nature?
Yes. Liposome-like structures occur naturally in many biological systems. One frequently cited example is milk, in which fats also occur as microscopic lipid structures surrounded by a layer of phospholipids. This shows that living organisms have long used similar mechanisms for transporting substances.
What should I look for when choosing a liposomal supplement?
When choosing a liposomal supplement, it is worth paying attention to several elements:
- Whether the manufacturer describes the technology in specific terms,
- Whether particle size information is available,
- Whether the formulation was tested by laboratory methods,
- Which phospholipids were used,
- Whether the product has a stable formulation in an aqueous environment.
The more transparent information about the technology, the easier it is to assess the actual quality of the product.
Does the name “liposomal” always mean advanced technology?
Not always. In practice, the term “liposomal” is sometimes used in a very broad way. Therefore, it is worth paying attention not only to the name of the product, but also to the technological data and laboratory tests confirming the structure and size of the particles.






